Machine Learning in QM/MM Molecular Dynamics Simulations of Condensed-Phase Systems | Journal of Chemical Theory and Computation

EiKO FTD Halogen Light Bulb (12-Pack), 12 Voltage Rating, 20 Watts, 1.67 Amps, GU4 Base, MR11 Bulb, C-8 Filament, 1.38"/35.0mm MOL, 1.39"/35.3mm MOD, 600 Approx Initial Max Beam CP

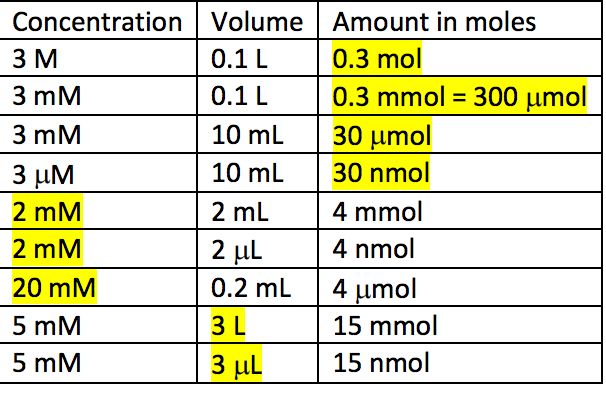
Molar Concentrations. Molarity is the number of moles of solute that can dissolve in 1 L of solution. Molar concentration (mol/L) = Amount of solute (mol) - ppt download
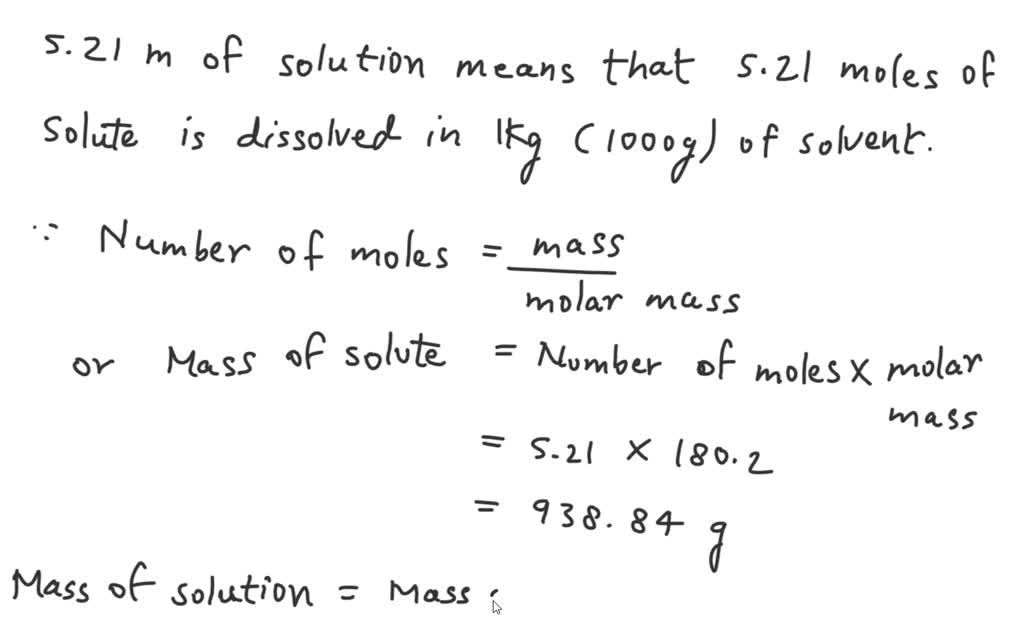
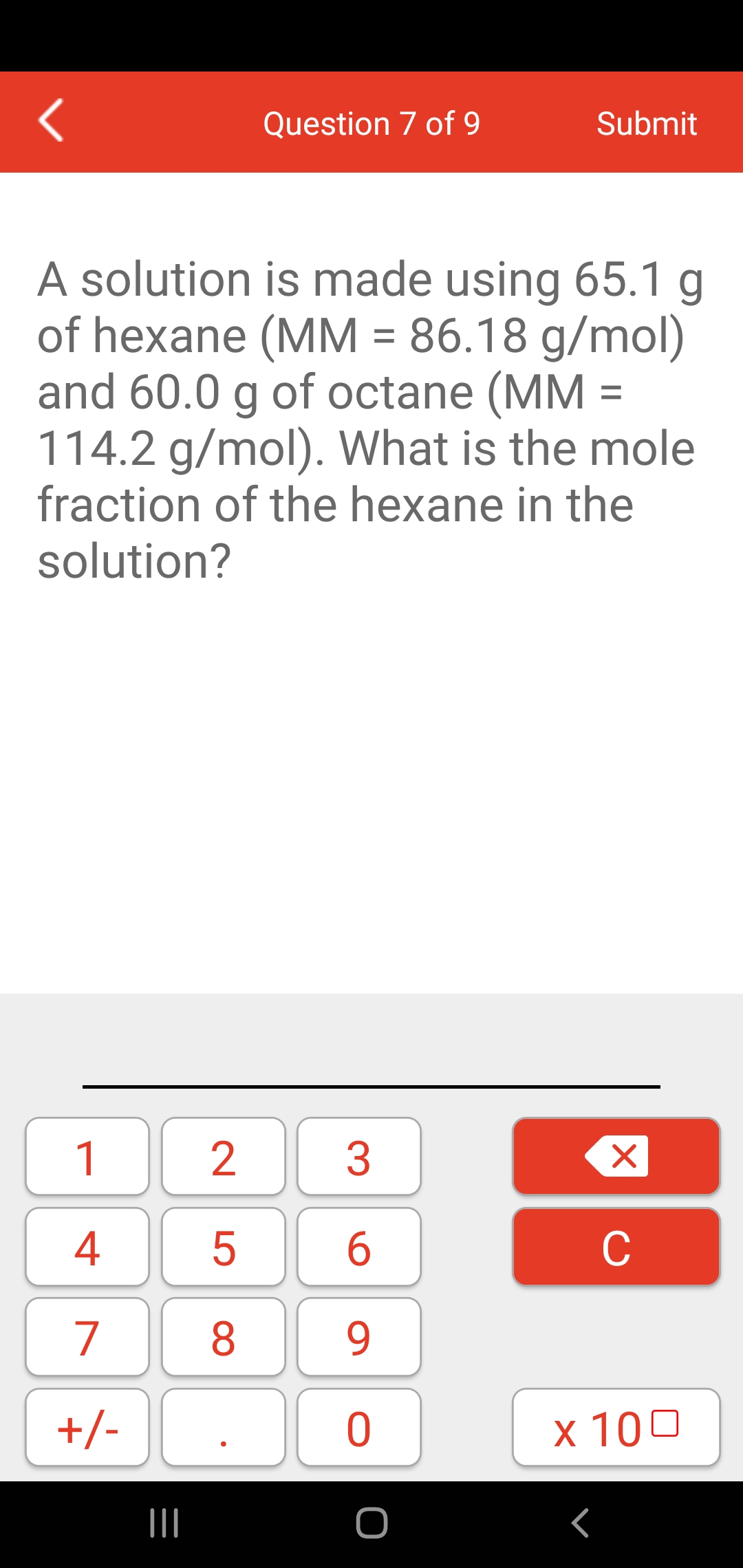
SOLVED: An aqueous solution of glucose (MM = 180.2 g/mol) has a molality of 5.21 m and a density of 1.20 g/mL. What is the molarity of glucose in the solution?

QM/MM Molecular Modeling Reveals Mechanism Insights into Flavin Peroxide Formation in Bacterial Luciferase | Journal of Chemical Information and Modeling